
Hebron students know (because we've all been preaching it for some time, now), attendance is so very important! You can't learn if you're not here, after all! And it appears to be sinking in and paying off because after winning the Bullitt County Attendance Trophy at the start of school and then losing it to Mt. Washington Middle, we've won it back again for our excellence attendance in the month of September! Way to go! Be sure to scope out that behemoth of a trophy in the front office, and keep making sure that you're here every day. We've been doing a fantastic job. Let's keep it up, and keep the trophy for October! And for my part, I'll do my best to keep well so I can be here everyday with you! Now, without further delay...

- We've colored blank Periodic Tables following these instructions to look something like this so we know the Atomic Families.
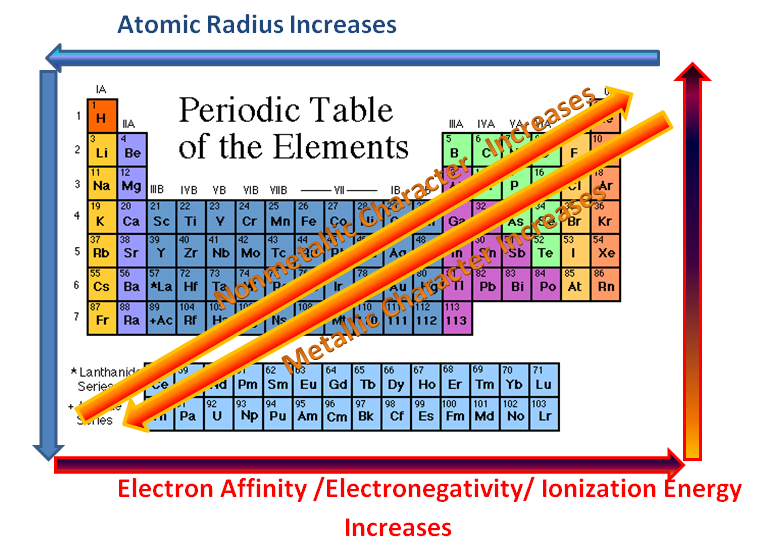
- We've colored other trends (or patterns) in the Periodic Table, such as this[1], or this[2], learning about the difference between metals, nonmetals, and metalloids (or "semi-metals"), as well as reactivity (i.e., which groups are the most chemically reactive).
- Classes got to watch these videos [1, 2, 3] of Alkali Metals reacting violently with water. Clearly, the further down a Group you go, the more reactive. Francium (Fr) is the most chemically reactive metal, and Fluorine (F) the most chemically reactive nonmetal. (Noble Gases, of course, are inert, meaning they do not react.)

- Francium is also the most "metallic" of the elements, as metallic properties increase when you move towards the bottom-left of the table. Meanwhile, when you move towards the upper, right-hand side of the table (towards Fluorine), the "non-metallic" properties of the elements increase (with semi-metals being the balance in between).
- Electron Affinity, Electronegativity, and Ionization Energy all follow the same trend. Electron Affinity is something like how tightly an atom clings to its electrons, or how reluctant it is to let any go. Ionization Energy is the amount of energy it would take to rip away an electron, and Electronegativity is something like how easily an atom can attract more electrons towards itself. Fluorine and Chlorine and the elements in the upper-right of the table all really want additional electrons to complete their outer shell, so they have high Electronegativity, high Ionization Energy, and high Electron Affinity. Meanwhile, Francium and the Alkali metals on the left-hand side (and especially towards the bottom) are very quick to give away electrons. They have very low Electronegativity, Ionization Energy, and Electron Affinity. These trends are summarized in the diagrams below:
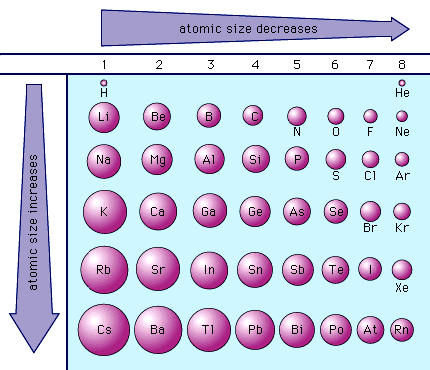
- The above diagram also refers to Atomic Radius, or how big an atom is. We graphed this data in class to develop a line graph that looked like this, identifying the Alkali Metals as those with the largest atomic radii (meaning Group 1 is the biggest). Meanwhile, atomic radius decreases as you go up a Group or across a Period. Therefore, Helium (He) in the upper, right-hand corner is the smallest. See the pattern for yourself below...

 RSS Feed
RSS Feed
