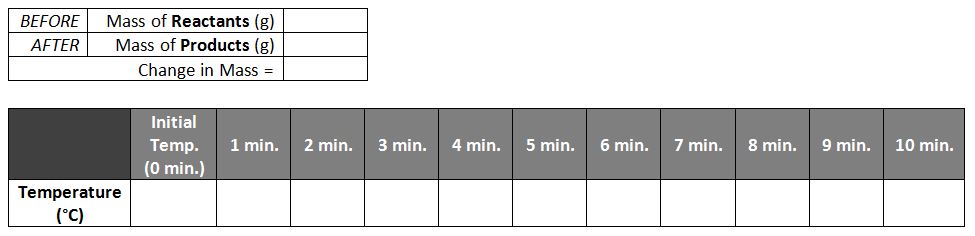
In this chemical reaction, iron combined with oxygen to form iron oxide (rust). We were particularly interested to see whether the mass would change as the two chemicals combined into one, and whether any thermal (heat) energy would be absorbed or given off.
First we washed steel wool cleaning pads in water to remove the soap, then soaked the steel wool in an acid solution (vinegar) to remove the outer protective coating. Then, we took an initial temperature reading using our thermometers and an initial mass with the triple-beam balance, recording both numbers in the data table below. Next, we wrapped the steel wool around the thermometer, inserted into a sealed flask, and recorded the temperature every 60 seconds for the next 10 minutes. Finally, we massed everything again to see whether mass was lost, gained, or remained the same, and graphed our temperature data to determine if the reaction was endothermic or exothermic.
Next, students performed their own exothermic reaction by combining hydrogen peroxide with live yeast. They measured the initial mass of the system, as well as the initial temperature, and compared them to the temperature and mass once the reaction had occurred. We concluded, as with the rusting demonstration, that the mass was the same (proving the Law of Conservation of Mass), and that in these instances the temperature had gone up, indicating exothermic reactions. Finally, students called the teacher over to conduct a flaming splint test, and observed that the smoldering wooden splint burst into flames when exposed to the gas inside the flask. From this observation, students inferred that the gas was likely oxygen, given our knowledge of the Fire Triangle, seen below.

 RSS Feed
RSS Feed
