[Check out the Resources page under "Chemistry" for links to some great Periodic Tables.]
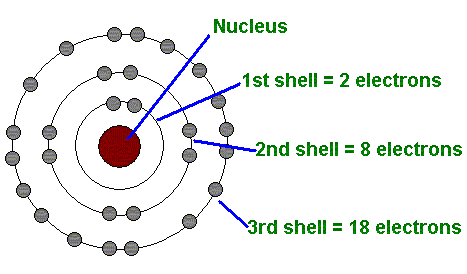
- Second, we know that the Periods in the Periodic Table tell us how many electron shells an atom has.
- Thirdly, we know the Groups in the Periodic Table can tell us how many valence electrons an atom has (or how many electrons there are in the outer-most shell).
We began discussing atomic Families, principally Group 18, known as the Noble Gases [see also Chem4Kids!]. These atoms have full valence shells, meaning the outer-most "ring" cannot possibly hold any more electrons. Adding another electron would mean adding a whole other shell, and they don't "want" to do that. They also don't "want" to give up any electrons due to the Octet Rule, or "Rule of 8," which says that every atom "wants" a full valence shell (8 electrons). Because Noble Gases don't want to give up any or get any new electrons, they are very "reluctant" to take part in chemical reactions. We call them "inert," meaning not chemically reactive.
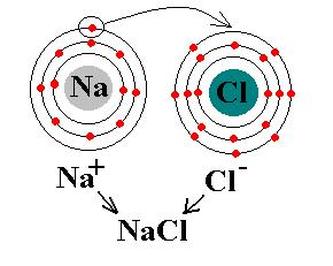
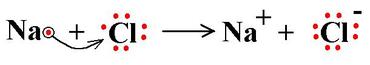
Meanwhile, Group 17 (Halogens, aka "salt makers") and Group 1 (Alkali Metals) are highly reactive, because each of them is so close to having a full valence shell! Halogens (Group 17=7 valence electrons) just need one more electron and they will have 8. Meanwhile, Alkaline Metals (Group 1) have this one extra electron that they just don't know what to do with! If it wasn't for that lone little bugger, they could drop that nearly empty electron shell and trim back down to a full 8 electrons in the previous shell, one "size" smaller.
So what do you think happens? You guessed it. The Group 17 atom with 7 valence electrons rips away that extra electron from the Group 1 atom and now, suddenly, it has 8. The Halogen atom has just turned into an ion and bonded with the Alkaline Metal.
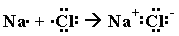

Carbon Dioxide is the same way. We'll practice looking at more chemical bonds tomorrow. (In the meanwhile, try some on your own!)
Also, remember that it's Friday tomorrow and we may very well be having one of our Friday Quizzes! So be prepared. (Review the practice quiz and activity from today!) :-)


 RSS Feed
RSS Feed
