Some classes moved ahead and illustrated the Lewis Dot Diagrams for the first 3 periods of the Periodic Table in order to observe the trends and patterns. [ Worksheet Here | Answers Here ].
|
Today we worked on turning our Bohr Models from Monday [worksheet here] into Lewis Models (also known as Electron Dot Diagrams). We began by practicing the identification of atoms by Period and Group [ chart 1 here | chart 2 here ] and reviewed that Period numbers indicate the number of electron orbitals, or shells, in an atom, and Group numbers tell us the number of valence electrons (electrons in the outermost shell).
Some classes moved ahead and illustrated the Lewis Dot Diagrams for the first 3 periods of the Periodic Table in order to observe the trends and patterns. [ Worksheet Here | Answers Here ].
0 Comments
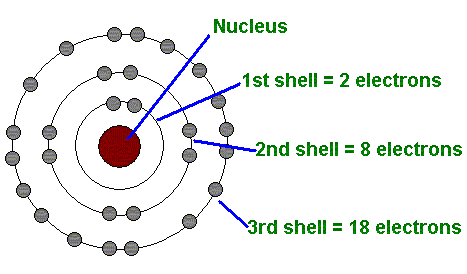
Today was Picture Day! In Science, we practiced drawing Bohr Models, which we originally learned about in class yesterday. Bohr Models depict an atom's electrons orbiting around its nucleus in specific energy levels, or shells. This slideshow presentation can show you how to draw the Bohr Models, and then we completed this simple practice worksheet.
Today we reviewed how to read a square on the Periodic Table to determine an atom's number of protons, neutrons, and electrons. We completed this worksheet, learning how to draw those electrons in their correct energy levels, or shells, when constructing a Bohr Model or a Lewis Dot Diagram. We will be practicing Bohr Models and Lewis Dot Diagrams, both, for the next couple of days.
Today was an Early Release Day. In the morning, in homeroom, we practiced an On-Demand Writing prompt that asked students to create an article for our Team Newsletters sharing a narrative of a time when they had shown H.E.A.R.T., and how that affected others around them. (Look for some fantastic student responses in upcoming Newsletters!) Then, in our shortened-period Science class, we completed this worksheet practicing how to find the number of protons, neutrons, and electrons from the information contained in the Periodic Table (specifically atomic number and atomic mass). The primary points to remember are: Atomic Number = # of Protons = # of Electrons (in a regular atom) Atomic Mass = # of Protons + # of Neutrons (consequently, # of Neutrons = Atomic Mass - Atomic Number) We'll practice again tomorrow. :-)
Today we focused on vocabulary. These two worksheets were pasted into our notebooks (pp. 30-31) and the definitions were matched with the terms. The vocab words are:
Additional notes for several of the words were copied from the board. For instance, students were asked to note that protons and neutrons each have a mass of 1 atomic mass unit (amu) or "dalton" apiece, while electrons have practically zero mass, thus resulting in an atomic mass equal to the sum of the protons plus neutrons.  Imagine trying to find the weight of all the Asian and African elephants in a zoo, plus the flies buzzing around their heads. The flies weigh practically nothing compared to the elephants! Same with electrons compared to protons and neutrons. Some students were still confused over the difference between neutrons and electrons, thinking that neutrons (beginning with the letter "n") were negative. I proposed that students think of it like this:
Get it? :-D |
Mr. FranklinWelcome to my blog! Archives
September 2015
Categories
All
|
 RSS Feed
RSS Feed
