|
| Students cut out and matched definitions to these vocabulary terms using their foldable notes booklet from yesterday as a resource/reference. Next, they classified a dozen or so real-world examples as being either elements, compounds, or mixtures (using the booklet's 3-column chart). |
|
Today students reviewed the 12 new vocabulary terms they were introduced to yesterday. Most classes got to view the StudyJams! video on Mixtures and conducted the practice self-test. Finally, students applied their terms to create a Flowchart illustrating the common divisions of matter.
0 Comments
Today students started off by gluing these notes into their Interactive Notebooks...
Next, we reviewed some older, foundational information on matter and then went through the new terms, one-by-one, highlighting key words and big ideas in each definition. Some classes watched the Scholastic StudyJam! on Elements & Compounds and/or conduct the BBC Bitesize Activity on Compounds & Mixtures. Each class then examined diagrams of Pure Elements, Compounds, Mixtures, etc. and created some of their own in the Table above. Afterwards, students worked cooperatively to identify each of the following diagrams accordingly. Finally, most classes were able to view the BrainPOP! Summary and complete a short 10-question Check-for-Understanding Formative Assessment.
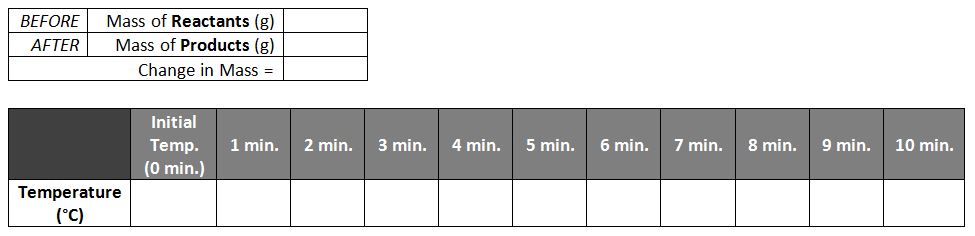
We started the week off with a demonstration of the "oxidation" of iron, a.k.a, rusting. In this chemical reaction, iron combined with oxygen to form iron oxide (rust). We were particularly interested to see whether the mass would change as the two chemicals combined into one, and whether any thermal (heat) energy would be absorbed or given off. First we washed steel wool cleaning pads in water to remove the soap, then soaked the steel wool in an acid solution (vinegar) to remove the outer protective coating. Then, we took an initial temperature reading using our thermometers and an initial mass with the triple-beam balance, recording both numbers in the data table below. Next, we wrapped the steel wool around the thermometer, inserted into a sealed flask, and recorded the temperature every 60 seconds for the next 10 minutes. Finally, we massed everything again to see whether mass was lost, gained, or remained the same, and graphed our temperature data to determine if the reaction was endothermic or exothermic. Afterwards, we removed the steel wool, now covered in rust, to make some qualitative observations. We noted color change, a distinct odor, and that heat had been produced -- all signs of a chemical change. Tomorrow we will compare to see whether the rusted pads have the same physical properties (like the ability to conduct electricity). Next, students performed their own exothermic reaction by combining hydrogen peroxide with live yeast. They measured the initial mass of the system, as well as the initial temperature, and compared them to the temperature and mass once the reaction had occurred. We concluded, as with the rusting demonstration, that the mass was the same (proving the Law of Conservation of Mass), and that in these instances the temperature had gone up, indicating exothermic reactions. Finally, students called the teacher over to conduct a flaming splint test, and observed that the smoldering wooden splint burst into flames when exposed to the gas inside the flask. From this observation, students inferred that the gas was likely oxygen, given our knowledge of the Fire Triangle, seen below. Today students began with a Warm-up Exercise that asked them to consider several examples of changes to matter, and asked whether the material or substance present at the end of each change was the same material or substance that was present at the beginning.
We utilized several interactives to help illustrate these changes: We afterwards watched a brief Scholastic Studyjams video on "Physical & Chemical Changes to Matter," and took the seven-question Practice Quiz. We constructed a T-chart in our Science Journals to summarize the major differences between physical and chemical changes, based upon what he had learned so far in from class discussion, interactives, and video. We then cut-out and glued in a chart that assisted us in classifying a number of changes to matter as either Physical or Chemical. |
Mr. FranklinWelcome to my blog! Archives
September 2015
Categories
All
|
 RSS Feed
RSS Feed
